
|
||||
| Home | Services | CDISC | Software | About us |
Now that Electronic Data Capture (EDC) is well established, and that most EDC vendors have embraced the CDISC ODM Standard, the next logical step is the integration of EDC systems with Hospital Information Systems and Electronic Health Records (EHR).
XML4Pharma is a very active member in a number of IHE-CDISC working groups that develops demos, prototypes and standards for EDC-EHR integration (IHE profiles RFD - Retrieve Form for Data Capture - and CRD - Clinical Research Data Capture) integration, and RPE - Retrieve Protocol for Execution).
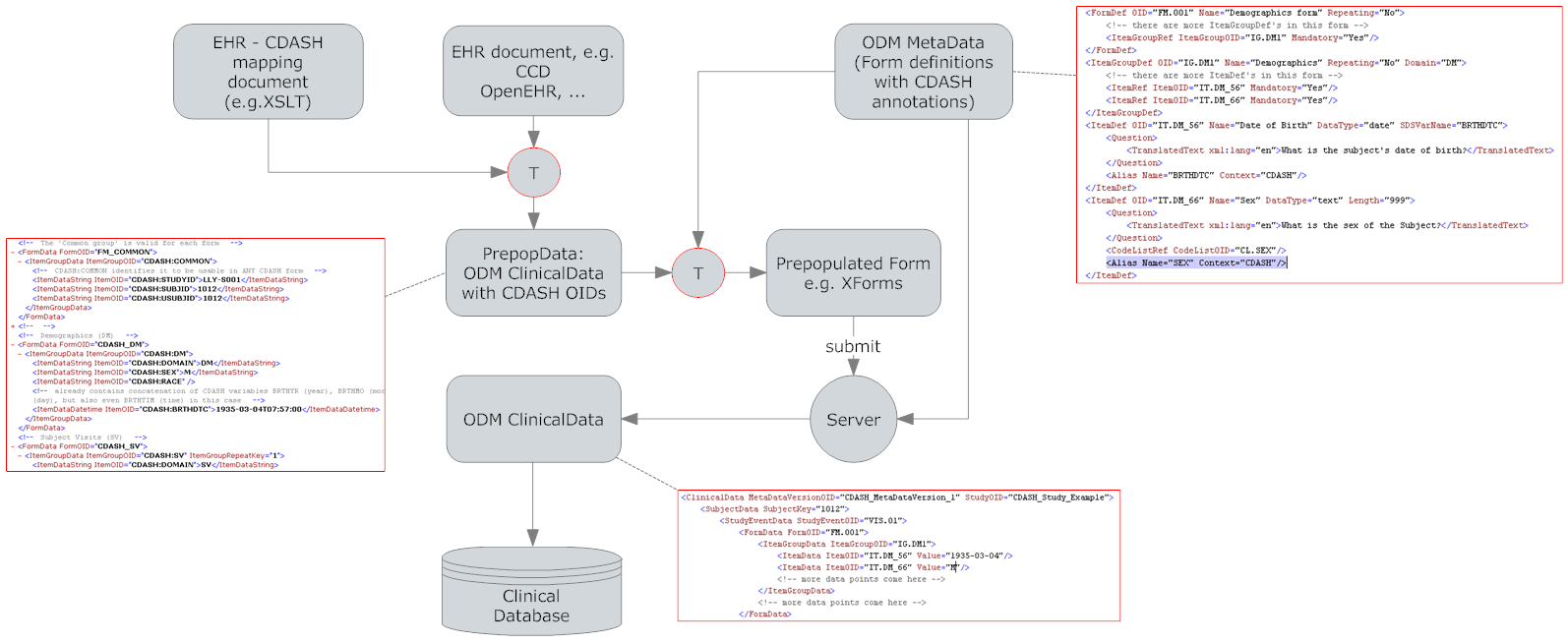
For the CRD initiative, we not only developed technology to transform EHR data from CCD (HL7 Continuity of Care Document) to an intermediate format
(so-called "PrepopData"), but also contributed considerably to the definition of the format of the PrepopData.
These PrepopData contains EHR data formatted as ODM 1.3 ClinicalData, which can be read by the EDC system, in order to prefill eCRFs (electronic Case Report Forms).
The great advantage of this integration is that the physician does not need to enter a good amount of patient information twice (once in the EHR system, and once in the EDC system), but can just have the EDC eCRFs prefilled with data from the EHR system.
Currently we are contributing to the RPE profile (Retrieve Protocol for Execution), where we are contributing to the further development of an ODM extension for trial design (ODM-TD), covering trial summary, machine-readable inclusion/exclusion Criteria, workflowing of trial activities, timing of activities etc.. We are also developing examples (ODM instance files) for testing and demonstrating the power of machine-readable protocols. Furthermore, we are investigating whether a mapping between the ODM-TD and the HL7-v3-XML messages being developed on behalf of the FDA is possible.
Results: CDASH-EHR integration
In first instance, the working group developed an interface between the CCD and the CDISC CDASH forms. These interfacing have been demonstrated at the IHE Connectathon in Chicago and in Vienna.
Although not in the IHE profile, we also developed stylesheets and technology to interface EHR and EDC for the case of OpenEHR, another well established (especially in Europe) EHR standard.
Our presentation "Integration of EHRs with CDISC, CDASH and ODM A European Initiative" given at the European CDISC Interchange in Budapest (April 2009) can be found here.
As one of the main contributors to the IHE-CDISC projects, we provide services to EDC and EHR vendors wishing to come to integration of healthcare and clinical research.
We also have a demo application available on our application server, showing how an EDC form can be started from within an hospital information system, and is prepopulated with data from the electronic health record of the patient.
